Chapter 8: Elimination Reactions
A nucleophilic substitution reaction is not the only possible reaction for alkyl halides and other substrates with a good leaving group. These substrates can also undergo elimination reactions. In an elimination reaction, a small molecule (XY) is removed from two adjacent atoms of the reactant, and a multiple bond is formed in the product:
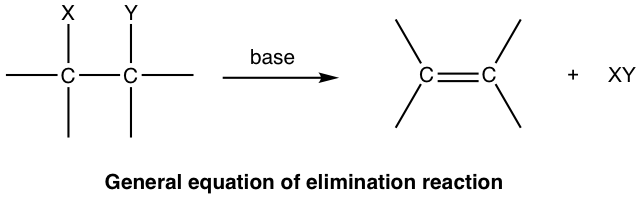
For an elimination reaction of alkyl halides, the major product alkene is produced together with the small HX (X is halogen) molecule, which is the side inorganic product. Such reaction with removal of a proton and a halide ion is called dehydrohalogenation. Dehydrohalogenation is a commonly applied method for the synthesis of alkene.
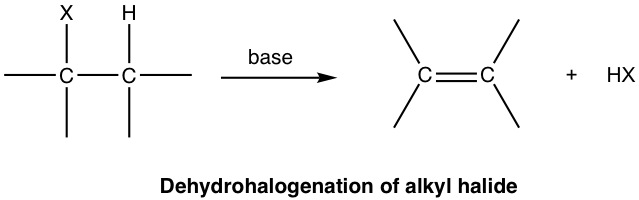
In the discussions of elimination reactions, the carbon atom that bonded to the leaving group (halogen for alkyl halide) is called the alpha (α) carbon atom, and the carbon atom adjacent to α-carbon is called the beta (β) carbon atom. In dehydrohalogenation, the hydrogen atom on β-carbon is eliminated together with halogen, as HX; therefore, the reaction is often called β-elimination or 1,2-elimination. (The number 1,2- indicates that the atoms being removed are on two adjacent carbons and not necessarily C1 and C2 atoms.)
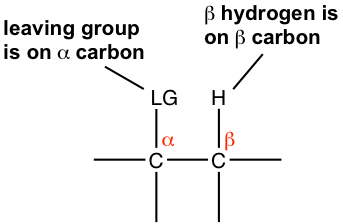
Beside the substrate, a base is required for elimination reactions other than the nucleophile for substitution. Similar to substitution reactions, elimination reactions also have different rate laws, and therefore involve different mechanisms. The elimination mechanisms are E1 and E2. You may expect that E1 is a unimolecular reaction with the first order rate law and E2 is the second order bimolecular reaction. That is correct. We will go through the mechanism in detail first, then compare between elimination and substitution.

