Chapter 3: Acids and Bases: Introduction to Organic Reaction Mechanism Introduction
Answers to Chapter 3 Practice Questions
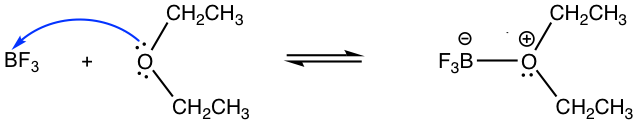
3.1 Predict and draw the products of the following reaction; use curved arrows to show the mechanism.
3.2 Practice drawing the resonance structures of the conjugate base of phenol by yourself!
Solutions are included in the section.
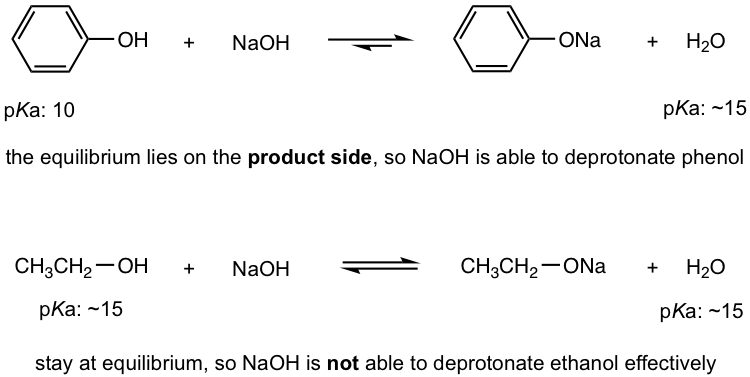
- It is the special acidity of phenol (and other aromatic alcohol) that allows NaOH to be used to deprotonate phenol effectively, but this is not the case for normal alcohols, like ethanol. Show the reaction equations of these reactions and explain the difference by applying the pKa values.
3.3 Show the product of the following LA-LB reaction: