Chapter 9: Free Radical Substitution Reaction of Alkanes
9.6 Synthesis of Target Molecules: Introduction to Retrosynthetic Analysis
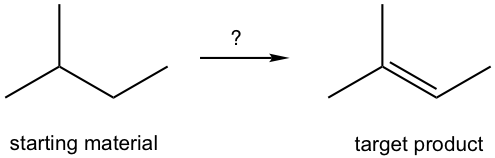
So far, we have learned three major types of reactions: nucleophilic substitution, elimination, and the halogenation of alkane (radical substitution). Now, we will see how to put the knowledge of these reactions together for application, that is, how to design a synthesis route for a target (desired) compound from available starting materials.
Building larger, complex organic molecules from smaller, simple molecules is the goal of organic synthesis. Organic synthesis has great importance for many reasons, from testing the newly developed reaction mechanism or method to replicating the molecules of living nature, to producing new molecules that have potential applications in energy, material, or medicinal fields.
It usually takes multiple steps, from a few to 20 or more, to synthesize a desired compound, and therefore it would be challenging to visualize from the start all the steps necessary. A common strategy for designing a synthesis is to work backward; that is, instead of looking at the starting material and deciding how to do the first step, we look at the product and decide how to do the last step. This process is called retrosynthetic analysis, a technique frequently applied in organic synthesis. We will introduce the basic ideas of retrosynthetic analysis here, and for practice purposes, the starting material will always be defined for our examples.
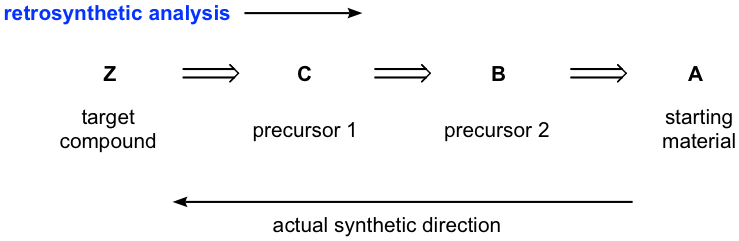
Retrosynthetic analysis can usually be shown in the above way, with the open arrows indicating that the analysis is backward. We first identify precursor 1 that could react in one step to make the target compound, then identify the next precursor that could react to give precursor 1, and repeat the process until we reach the starting material. Please note that the analysis is done in this way to show the “thinking or ideas” for solving the problem, so typically the reagents/conditions required for each step are not specified until the synthesis route is written in the forward direction. Also, you may come up with multiple routes sometimes, with different precursors, and then the most efficient synthesis route can be determined by evaluating the possible benefits and disadvantages of each path.
Examples
Design the synthesis route of methoxymethylbenzene starting from toluene.
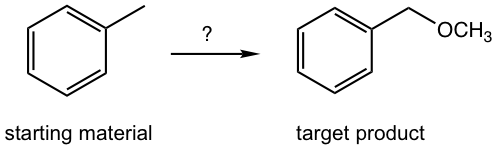
Approach: The target compound is an ether. We have learned that the SN2 reaction is a reasonable way to introduce different functional groups by applying different nucleophiles (section 7.3), that said the reaction between CH3O– (nucleophile) and halide gives the desired ether, and the halide can be the “precursor 1”. The halide precursor can then be directly connected with the starting material, toluene, through the halogenation that we just learned in this chapter. This is an easy example that only involves two steps.
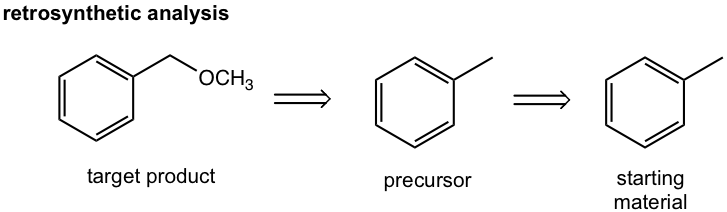
Solutions:
The analysis can then be transferred to the solution of the question by showing the reactions in forward direction and include the reagents/condition for each step.
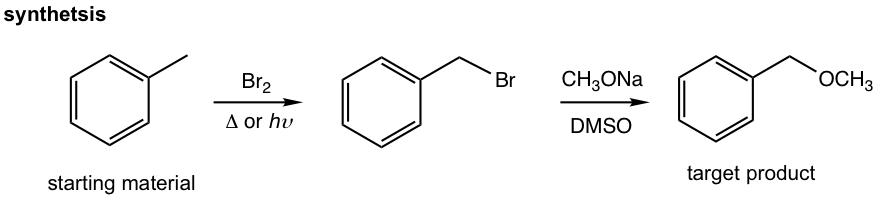
Synthesis route design is a challenging topic that requires a lot of practice. To do it well, you should be very familiar with all types of reactions in terms of how the functional groups are transformed and what reagents and conditions are involved. Sometimes, some reaction features, like stereochemistry, will be useful as well.
Exercises 9.4 Design the synthesis route.