Chapter 7: Nucleophilic Substitution Reactions
7.2 SN2 Reaction Mechanisms, Energy Diagram and Stereochemistry
SN2 Reaction Mechanism
Let’s still take the reaction between CH3Br and OH– as an example of an SN2 mechanism.
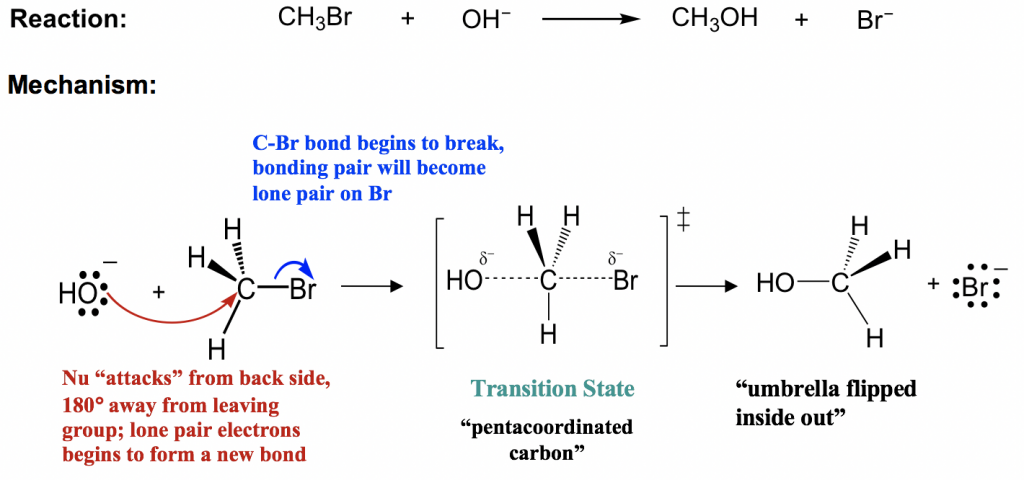
An SN2 mechanism involves two electron pair transfers that occur at the same time; nucleophile attacking (red arrow) and leave group leaving (blue arrow). The nucleophile OH– approaches the electrophilic carbon from the back side, the side that is opposite to the direction that the leaving group Br leaves. With the nucleophile OH– getting closer, the Br starts to leave as well. The new C—OH bond formation and the old C—Br bond breaking occur at the same time. In a very short transient moment, the carbon atom is partially connected with both OH and Br, which gives the highest energy level state of the whole process called the transition state. In the transition state of an SN2 reaction, there are five groups around the carbon and the carbon can be called “pentacoordinated”. As the OH– continues to get closer to the carbon, the Br moves further away from it with the bonding electron pair. Eventually, the new bond is completely formed and the old bond is completely broken, which gives the product CH3OH.
In the mechanism, the reaction proceeds in a single step that involves both the nucleophile and the substrate, so increasing the concentration of either increases the possibility of a collision, which explains the second-order kinetics of an SN2 reaction. With both nucleophile attacking and leaving group leaving happen at the same time, SN2 is also said to be a concerted mechanism, as concerted means simultaneous.
Notes for drawing an SN2 mechanism:
- The two arrows must be shown when drawing the SN2 mechanism. Both have to be shown with the proper direction: nucleophile attack from the direction that is opposite to the leaving group leaves, i.e., backside attack.
- The transition state is optional (depending on the requirement of the question). However, it is important to understand that the reaction process goes through the transition state before producing the products.
- Please pay attention to the fact that for the product, the positions of the three hydrogens around the carbon are all pushed to the other side, and the overall configuration of the carbon gets inverted, like an umbrella flipped inside out in a windstorm. It seems to not really matter for the product (CH3OH) ) in this reaction, but it does make a difference if the carbon is a chirality center.
Energy Diagram of the SN2 Mechanism
The energy changes for the above reaction can be represented in the energy diagram shown in Fig. 7.2b. SN2 is a single-step reaction, so the diagram has only one curve. The products CH3OH and Br– are in lower energy than the reactants CH3Br and OH–, which indicates that the overall reaction is exothermic and the products are more stable.
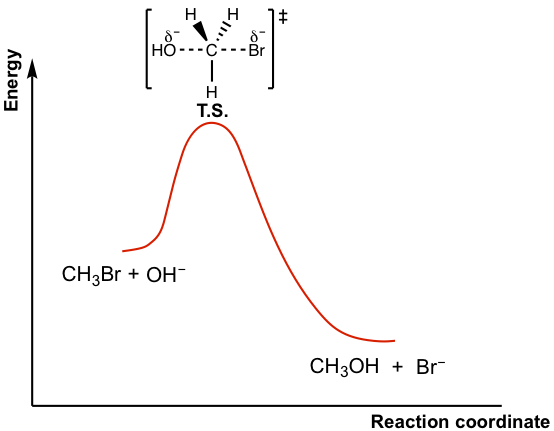
The top of the curve corresponds to the transition state, which is the highest-energy structure involved in the reaction. A transition state always involves partial bonds, partially formed bonds and partially broken bonds, and therefore it is very unstable with no appreciable lifetime. The transition state therefore can never be isolated. The structure of the transition states is usually shown in a square bracket with a double-dagger superscript.
The Effect of Alkyl Halide Structure on SN2 the Reaction Rate
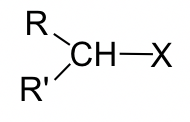
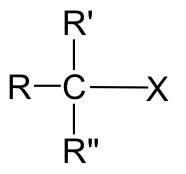
For the discussions on the SN2 mechanism so far, we focused on the reaction of methylbromide CH3Br. Other alkyl halides could undergo SN2 reactions as well. The studies on the reaction rate for SN2 indicate that the structure category of electrophilic carbon in alkyl halide dramatically affects the reaction rate.
|
Type of Alkyl Halide
|
Alkyl Halide Structure |
Relative Rate |
| Methyl |
CH3X |
30 |
| Primary (1°) |
RCH2–X
|
1 |
| Secondary (2°) |
|
0.03 |
| Tertiary (3°)
(no SN2 reaction) |
|
negligible |
Table 7.1 Relative Reaction Rate of SN2 for Different Types of Alkyl Halide
As shown in Table 7.1, methyl and primary halides are the substrates with the highest rate, the rate decreases a lot for secondary halides, and the tertiary halides do not undergo an SN2 reaction at all because the rate is too low to be practical.
The relative reactivity of alkyl halides towards SN2 reaction can therefore be summarized as:

Why is the trend like this? This can be explained by the mechanism of the SN2 reaction. Actually, this is among the experimental evidence scientists used for proposing the mechanism. A key feature in the SN2 mechanism is that the nucleophile attacks from the back side. When nucleophiles approach the carbon, it is easiest to get close to the methyl carbon because the hydrogen atoms connected to carbon are small in size. With the size of the groups connected to the carbon getting larger, it becomes more difficult to access the carbon, and such an approach is completely blocked for tertiary carbons with three bulky alkyl groups connected. Therefore, the reactivity difference is essentially caused by the steric effect. The steric effect is based on the steric size or volume of a group. Because of the steric hindrance of bulky groups on the electrophilic carbon, it is less accessible for nucleophiles to do back-side attacks, so the SN2 reaction rate of secondary (2°) and tertiary (3°) substrates dramatically decreases. The 3° substrates never go with the SN2 reaction mechanism because the reaction rate is too slow.

The Stereochemistry of SN2 Reaction
Another feature of the SN2 reaction mechanism is that the overall configuration of the carbon in the product gets inverted compared to that of the reactant, as an umbrella flipped inside out. Such inversion of the configuration is called Walden inversion. Let’s see what are the stereochemistry consequences for such inversion.

Start with the (R)-2-bromobutane. The SN2 reaction produces only one enantiomer of the 2-butanol product, and it is predictable that the configuration of the product is supposed to be S because of the configuration inversion.
Exercises 7.1
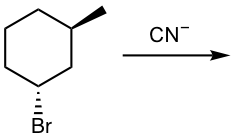
Show the product of the following SN2 reaction (CN– is the nucleophile):