Chapter 5: Stereochemistry
5.5 Fisher Projection
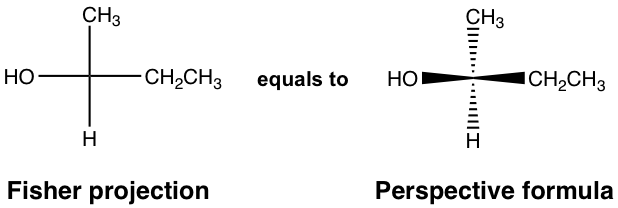
For the discussions so far, the perspective formula with solid and dashed wedges has been used to represent the 3D arrangement of groups bonded to a chirality center. In addition, there is another broadly applied formula for that purpose: the Fisher projection. A Fisher projection is a shortcut for showing the spatial group arrangement of a chirality center; it is easier to draw and recognize and is particularly useful for showing structures with more than one chirality center.
In a Fisher projection, the chirality center is shown as the intersection of two perpendicular lines. The horizontal lines represent the bonds that point out of the plane, and the vertical lines represent the bonds that point behind the plane.
Assigning an R/S Configuration in a Fisher projection
Taking the following compound as an example:
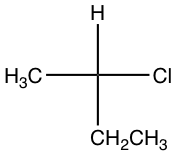
- Assign group priority as we usually do.
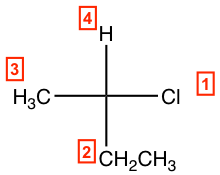
- If the lowest priority group (#4 group) is on a vertical bond, determine the priority decrease direction from #1→#2→#3 as usual to get the configuration; clockwise is R and counterclockwise is S.
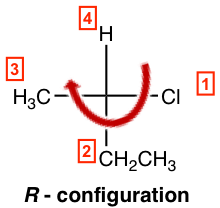
So, the example here is an R-isomer, and the complete name of the compound is (R)-2-chlorobutane.
- If the lowest priority group is on a horizontal bond (as is the case in the following structure), determine the priority decrease direction as in step 2, then reverse the answer to the opposite way to get the final configuration.

So, the example here is a S-isomer, and the complete name of the compound is (S)-2-chlorobutane.
Exercises 5.6
Explain why in step 3 of the above procedure, the answer should be reversed to get the final (actual) configuration.
Answers to Chapter 5 Practice Questions
Exercises 5.7: Indicate the configuration of the following structures.
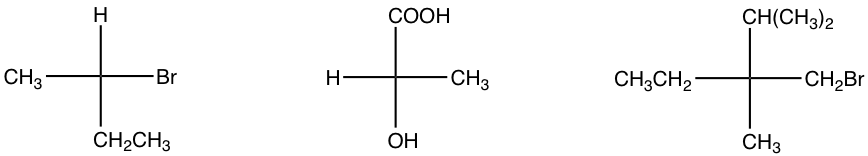
Properties of a Fisher projection:
1. One switch (interchange) of two groups in a Fisher projection inverts the configuration, and two switches bring the original isomer back.
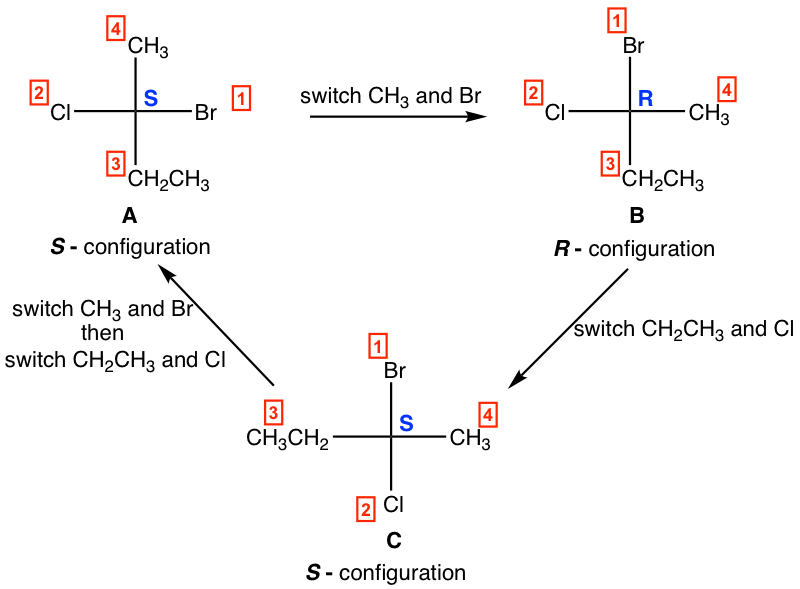
For the above structures:
- one switch of A leads to B, and A and B are enantiomers.
- one switch of B leads to C, and B and C are enantiomers.
- two switches of C lead to A, and A and C are identical.
2. Rotate the Fisher projection 180º to get the same structure, with the configuration retained.
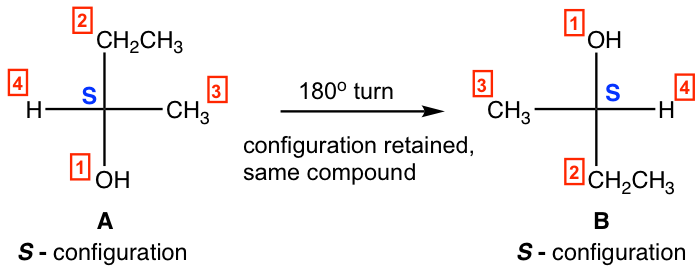
- A 180º rotation of A leads to B, and A and B are identical.
3. Rotate the Fisher projection 90º to get the configuration inverted.

- 90º rotation of A leads to B, and A and B are enantiomers.
Do NOT rotate the Fisher projection 90º, unless you have to. Keep in mind that the configuration gets inverted by a 90º rotation.

